Introduction
Allergic rhinitis is an allergic inflammation of the nasal mucosa induced by pollen, house dust mites or other allergens, whose symptoms include nasal congestion, sneezing, nasal pruritus and increased nasal discharge [1, 2]. The morbidity of allergic rhinitis is related to the season, and prevalence has been increasing in the past few years [3, 4]. Mast cells play an important role in orchestrating IgE-mediated allergic responses. After exposure to allergen, mast cells are activated and release preformed mediators such as histamine, heparin, serine proteases, and chemokines, promoting the occurrence of allergic inflammation [5-8]. Therefore, the study of the mechanism of mast cells mediating allergic rhinitis might provide a new way to alleviate allergic rhinitis.
Paeoniflorin (PF) is an organic compound extracted from the Chinese herb Paeonia lactiflora Pall., and much research has reported that PF has a significant effect on the treatment of tumors, neurological disease, and depression [9-14]. In recent years, PF has been proven to regulate inflammation and balance the subsets of immune cells to ameliorate multiple diseases, such as arthritis, asthma, and diabetic wound healing [15-18]. These results suggest that PF has a good anti-allergic effect. However, the biological functions of mast cells modulated by PF in allergic rhinitis are rarely reported.
Therefore, in this study, we used dinitrophenol (DNP)- human serum albumin (DNP-HSA) to stimulate mast cells to establish cell models and then investigated the anti- allergic effect of PF. We speculated that PF treatment might inhibit the degranulation of mast cells, which may provide a fresh idea for PF treatment for allergic rhinitis.
Material and methods
Cell culture and treatment
Rat basophil leukemia cells (RBL-2H3) were provided by the Cell Bank of the Chinese Academy of Sciences (Shanghai, China), and rat peritoneal mast cells (RPMCs) were provided by BioVector Science Lab (Beijing, China). In accordance with guidelines, RBL-2H3 cells and RPMCs were cultured in Dulbecco’s Modified Eagle Medium (DMEM; Thermo Scientific, Waltham, MA, USA) supplemented with 15% fetal bovine serum (FBS, Thermo Scientific) at 37oC with 5% CO2, and then incubated with 200 ng/ml DNP-specific IgE (Sigma, St. Louis, MO, USA) overnight. Next, RBL-2H3 cells and RPMCs were incubated with different doses of PF (purity: ≥ 98%; Merck; Fig. 1A) (0, 0.1, 1, 10, and 100 µM) for 1 h. After that, DNP-HSA (Merck, Darmstadt, Germany) was used to stimulate cell degranulation. The cells with or without PF treatment were stimulated with 20 ng/ml DNP-HSA for 4 h. Cells exposed to 10 µM dexamethasone (DEXA; Merck) for 24 h were used as the positive control. To activate the cGAS/STING signaling pathway, 100 µg/ml 5,6-imethylxanthenone-4-acetic acid (DMXAA, Merck) was used to stimulate RBL-2H3 cells and RPMCs for 2 h.
Fig. 1
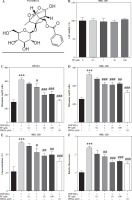
Effects of paeoniflorin (PF) on mast cell degranulation. A) Chemical structure of PF. B) MTT assay was applied to measure the viability of RBL-2H3 cells treated with different concentrations of PF (0, 0.1, 1, 10, 100 μM). C, D) The release of histamine in RPMCs and RBL-2H3 cells treated with or without PF (0.1, 1, 10 and 100 μM). 10 μM DEXA was used as the positive control. The release of E) β-hexosaminidase and F) intracellular calcium levels in RBL-2H3 cells treated with or without PF (0.1, 1, 10 and 100 μM). ***p < 0.001 vs. the control group. #p < 0.05, ##p < 0.01, and ###p < 0.001 vs. the DNP-HSA group
Cell viability assay
Cell viability was measured by a methyl thiazolyl tetrazolium (MTT) assay kit (Thermo Scientific). In brief, RBL-2H3 cells were incubated in 96-well plates at a density of 1 × 104 cells/well, and treated with different concentrations of PF (0, 0.1, 1, 10, and 100 µM) for 24 h. Then, the cells were incubated for 2 h with 1 mg/ml MTT at 37oC. Finally, the formazan crystal byproducts in the cells were dissolved by adding 100 µl dimethyl sulfoxide (DMSO) to each well. A microplate reader was used to measure the absorbance at 570 nm.
Histamine assay
Histamine was measured by a histamine enzyme-linked immunosorbent assay kit (ELISA; Sangon Biotech, Shanghai, China). In short, the cells were centrifuged at 4oC at 1000 ×g for 20 min to obtain the supernatant. The supernatant (50 µl) was incubated with 50 µl of biotin-conjugated antibody working solution for 45 min at 37oC. Subsequently, 100 µl of horseradish peroxidase (HRP)-conjugated streptavidin working solution was added to incubate for 30 min at 37oC. Then, 90 µl of substrate reagent was incubated with the complex for 15 min at 37oC. The reaction was stopped after adding 50 µl of stop solution. The optical density (OD) value was measured at a wavelength of 450 nm.
β-hexosaminidase assay
β-hexosaminidase was measured using a β-hexosaminidase activity assay kit (Cell Biolabs, San Diego, CA, USA). After centrifuging at 4oC at 1000 ×g for 20 min, the supernatant (50 µl) was collected and added to the 96-well microtiter black plate with β-hexosaminidase positive control. Then, 1× substrate (50 µl) was added to each well and incubated at 37oC for 15 min, protected from light. Finally, 1× neutralization buffer (100 µl) was added to each well. The plate was read at an excitation wavelength of 365 nm and an emission wavelength of 450 nm using a microplate fluorometer.
Determination of intracellular calcium level
The concentration of intracellular calcium was measured using the fluorescent indicator Fluo-3/AM (Invitrogen, Carlsbad, CA, USA). In brief, cells were preincubated with Fluo-3/AM (5 µM) for 1 h at 37oC, and then treated with PF and DNP-HSA. The fluorescence intensity was measured using a fluorescence plate reader at an excitation wavelength of 485 nm and an emission wavelength of 510 nm. The intracellular calcium levels were compared to those of untreated control cells set to 1 relative absorbance unit.
Western blot assay
Total protein was isolated by radioimmunoprecipitation assay (RIPA) lysis buffer (Thermo Scientific, USA). Protein samples were loaded onto 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) gel and then transferred to polyvinylidene fluoride (PVDF) membranes. Afterwards, the membrane was incubated with primary antibodies overnight at 4oC, including anti- p-JAK2 (ab32101, 1 : 1000), anti-JAK2 (ab108596, 1 : 1000), anti-p-STAT3 (ab267373, 1 : 1000), anti-STAT3 (ab68153, 1 : 1000), anti-p-AKT (ab38449, 1 : 1000), anti- cGAS (ab302617, 1 : 1000), anti-STING (ab239074, 1 : 1000), anti-p-p65 (ab76302, 1 : 1000), anti-p65 (ab32536, 1 : 1000), anti-GAPDH (ab9485, 1 : 2500) (all bought from Abcam, Cambridge, UK), and anti-AKT (#9272 1 : 1000) (Cell Signaling, Danfoss, MA, USA). After incubation with secondary antibody (Abcam, ab6721, 1 : 2000) at room temperature for 2 h, blots were visualized and photographed with an optical luminescence instrument (GE, USA).
Molecular docking
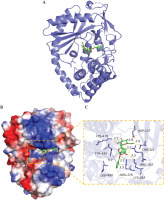
The 2D structure of PF was obtained from the PubChem database, and the 3D structure of the target protein was obtained from the PDB database. PyMOL software was used to remove water molecules and small molecule ligands in the protein structure, and then imported into AutoDock Tools 1.5.6 to hydrogenate the protein. Vina was used to complete molecular docking, and PyMOL was used to visualize the drawing.
Statistical analysis
Data processing was completed with SPSS 22.0 software. All results were represented as the mean ± standard deviation from three replicated experiments. Comparisons among multiple groups were implemented with one-way analysis of variance (ANOVA). P < 0.05 was considered statistically significant.
Results
Effects of PF on degranulation of mast cells
It is reported that PF has a great effect on inhibiting allergy [19]. In order to detect the effect of PF treatment on mast cells, we used PF in 0, 0.1, 1, 10, and 100 µM concentrations to treat RBL-2H3 cells. Then, we measured the influence of PF on RBL-2H3 cell viability and found that PF showed no effects on it (Fig. 1B). This meant PF exhibited no cytotoxicity. After that, 0.1, 1, 10 and 100 µM PF were treated on RBL-2H3 cells and RPMCs to measure the release of histamine, β-hexosaminidase, and intracellular calcium levels. These three mediators were all prominently elevated due to DNP-HSA treatment, whereas PF (1, 10 and 100 µM) and DEXA reversed the release of histamine and β-hexosaminidase, and decreased intracellular calcium levels (Fig. 1C-F). To sum up, PF alleviated mast cell degranulation induced by DNB-HSA. Since the recovery effect of 10 and 100 µM PF was closest to positive control DEXA, and the dose of 10 µM PF was lower, we chose 10 µM PF for subsequent experiments.
PF inhibits the activation of the cGAS/STING signaling pathway caused by DNP-HSA
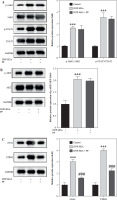
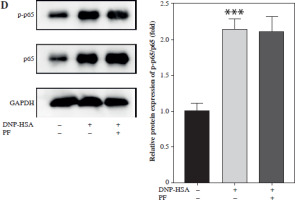
Numerous studies have shown that many signaling pathways were involved in mediating inflammation, such as the JAK/STAT, PI3K/Akt, NF-κB, and cGAS/STING pathways [20-27]. Therefore, we chose these pathways to identify whether they were involved in the regulation of mast cell degranulation by PF. The results showed that the protein levels of p-JAK2, p-STAT3, p-AKT, cGAS, STING, and p-p65 were increased by DNP-HSA, indicating that DNP-HSA activated the JAK2/STAT3, AKT, cGAS/STING and NF-κB signaling pathways (Fig. 2A-D). We found that only the protein levels of cGAS and STING were downregulated by PF (Fig. 2C), suggesting that PF inactivated the cGAS/STING pathway induced by DNP-HSA. Afterwards, we performed molecular docking of PF with the target protein cGAS, and the results showed a good combination and high degree of matching (Fig. 3A-C). In conclusion, we demonstrated that PF may suppress activation of the cGAS/STING signaling pathway by targeting the expression of cGAS protein through binding to cGAS.
DMXAA can reverse the effect of PF on degranulation of mast cells stimulated with DNP-HSA
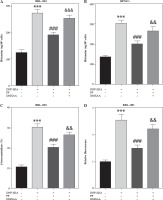
To verify whether PF is involved in the degranulation of mast cells by inhibiting the cGAS/STING pathway, we used the STING agonist DMXAA to activate this signaling pathway. The results showed that DMXAA increased the release of histamine and β-hexosaminidase and elevated the intracellular calcium level in mast cells treated with DNP-HSA and PF (Fig. 4A-D). Hence, we confirmed that PF regulated the degranulation of mast cells with DNP-HSA treatment through inhibiting the cGAS/STING signaling pathway.
Fig. 4
DMXAA reverses the inhibitory effect of paeoniflorin (PF) on degranulation in RBL-2H3 cells and RPMCs. A, B) RBL-2H3 cells and RPMCs were treated with DNP-HAS, PF, and DMXAA, and the release of histamine was detected using a histamine ELISA assay kit. C, D) The release of β-hexosaminidase and intracellular calcium level in RBL-2H3 cells. ***p < 0.001 vs. control group. ###p < 0.001 vs. DNP-HSA group. &&&p < 0.001 and &&p < 0.01 vs. DNP-HAS + PF group
Discussion
In this study, mast cells induced with DNP-HSA were used to investigate the degranulation effect of PF. Our results showed that PF reduced the release of histamine and β-hexosaminidase and decreased the intracellular calcium level in mast cells stimulated by DNP-HSA. In addition, we illustrated that PF inhibited the activation of the cGAS/STING signaling pathway.
Mast cells are primary effectors in allergic reactions, and they play an important role in diseases by releasing various mediators [28]. Histamine is one of the most representative mediators that has an effect on the cardiovascular system, extravascular smooth muscle, and gastric secretion [29]. Histamine nasal insufflation induces nasal itching, sneezing, and rhinorrhoea as well as nasal blockage [30]. It is reported that antagonizing IL-4 and IL-13 in vitro and anti-IL-4 treatment in mice prevented epithelial barrier disruption induced by histamine during the early phase of the allergic immune response [31]. β-hexosaminidase, a lysosomal enzyme released from mast cells, is another important mediator [32]. Cheong et al. [33] reported the inhibitory effect of hydroxystilbenes on the release of β-hexosaminidase, thus exerting anti-allergic action. Measurement of histamine and β-hexosaminidase is a way to observe the degranulation of mast cells, which could be a method to evaluate allergy condition.
Some traditional Chinese medicines have been proven to have a good therapeutic effect on allergic inflammation. For example, Kang et al. [34] verified that nothofagin suppresses mast cell-mediated allergic inflammation through a passive cutaneous anaphylaxis mouse model. Also, PF plays an important role in allergic disease [35, 36]. Wang et al. [37] reported that PF regulates the balance between inflammatory and anti-inflammatory cytokines to inhibit inflammatory responses in mice with allergic contact dermatitis. Sun et al. [38] proved that PF displays anti-inflammatory effects in the ovalbumin-induced asthmatic model by decreasing the expression of IL-5, IL-13, IL-17, and eotaxin. In this study, we investigated the anti-allergic function of PF in allergic rhinitis. Our data showed that PF significantly decreased the release of histamine and β-hexosaminidase in RBL-2H3 cells induced by DNB-HSA, suggesting that PF inhibits the degranulation of mast cells to attenuate allergy.
Intracellular calcium level is regarded as an important secondary messenger to trigger histamine release [39]. Wang et al. [40] reported PF reduced C48/80-induced calcium influx and suppressed mast cell degranulation to inhibit allergic response. Our study demonstrated that PF reversed the elevation of intracellular calcium level caused by DNB-HSA. Therefore, the suppressive effects of PF on mast cell degranulation might be through reducing the intracellular calcium level. All these results indicate that PF has is effective in inhibiting degranulation to relieve mast cell-mediated allergic inflammation.
Moreover, we found that PF inhibited the activation of the cGAS/STING signaling pathway in RBL-2H3 cells induced by DNP-HSA. In recent years, cGAS/STING has played an important role in immunotherapy and biotherapy [41, 42]. Some studies have also reported that the cGAS/STING pathway may promote inflammation [43-45]. Wang et al. [46] illustrated that PF attenuated the mast cell-mediated allergic inflammatory response by suppressing the NF-κB and MAPK signaling pathways; however, whether PF could participate in mast cell phenotypes by regulating the cGAS/STING pathway remains unknown. Our results also revealed effective combination of PF and the target protein cGAS, suggesting that PF binds to cGAS to inactivate the cGAS/STING pathway. Moreover, the STING agonist DMXAA could reverse the inhibition of mast cell degranulation caused by PF. These data imply that PF inhibits the mast cell-mediated allergic response through inhibiting the cGAS/STING signaling pathway.
However, the present study has limitations. The major limitation is that we only used a cell model to identify the anti-allergic function of PF in DNP-HSA-induced mast cells, which might be verified using animal experiments in our future work.
In conclusion, our study reveals that PF inhibits mast cell degranulation by suppressing the cGAS/STING signaling pathway, suggesting that PF may be a potential therapeutic drug for allergic inflammation mediated by mast cells.