Introduction
Immunoglobulin A (IgA) vasculitis (IgAV) is the most common form of vasculitis in the pediatric population. According to global registers, it is estimated that the prevalence among children ranges between 3 and 26,7 cases per 100,000 people [1-3]. Based on the 2008 IgAV classification criteria adopted by EULAR (the European League Against Rheumatism), PRINTO (the Paediatric Rheumatology International Trials Organisation) and PRES (the Paediatric Rheumatology European Society), a mandatory/obligatory criterion to diagnose IgAV is palpable purpura with at least one of the following symptoms: abdominal pain, typical histopathological changes, arthritis or arthralgia, renal involvement (hematuria and/or proteinuria) and IgA deposition in biopsy specimens (Table 1) [4-6]. From 30% to 60% of children with IgAV are affected by the accumulation of deposits (e.g. IgA) in glomeruli and the development of nephropathy in the course of IgA vasculitis with nephritis (IgAVN) [7]. In most cases, the disease is self-limiting, although 2-20% of the patients develop end-stage renal disease [2, 8]. The mild clinical course applies to patients with erythrocyturia and/or mild proteinuria (< 1 g/day, and UP : UC ratio < 100 mg/mmol); however, in around 20% of the patients with renal involvement, the disease manifests as nephrotic or nephritic syndrome [9, 10]. It may also present as impaired renal function, which increases the risk of progression to end-stage renal disease, which was detected in 1-7% of the cases [11-13].
Table 1
Classification criteria for immunoglobulin A (IgA) vasculitis (their fulfilment classifies this form of vasculitis with a sensitivity of 100% and a specificity of 87% [5]
Old age and low glomerular filtration rate (GFR) are well-known risk factors for the unfavorable course of IgAVN [12, 14]; however, researchers have not reached a consensus on these conclusions and in publications of Kawasaki et al. and Mir et al. did not observe this correlation [15, 16]. According to the latest meta-analysis gender is not significantly associated with poor outcomes in IgAVN [12], whereas a higher risk of developing IgAVN among patients with IgAV is found in boys [17]. In terms of urine changes, presence of nephrotic and nephritic- nephrotic syndrome is associated with a worse prognosis, whereas hematuria and mild proteinuria with/without hematuria have good clinical outcomes. Nephrotic syndrome persisting over three months was particularly related to poor prognosis. Also, the nephritic-nephrotic syndrome is an independent factor associated with unfavorable outcomes, whereas acute nephritic syndrome does not show this correlation [12].
As before, the gold standard of diagnosis and severity assessment of IgAVN is a kidney biopsy [10, 16]. For the evaluation of histopathological findings in the kidney, the Oxford classification, which so far has not been validated for IgAVN but has for IgA nephropathy (IgAN), is increasingly used [13, 18]. This scoring system includes five histologic parameters which form the MEST-C score: M – mesangial hypercellularity score (M0 ≤ 50% glomeruli, M1 > 50% glomeruli); E – the presence of endocapillary proliferation (E = 0: absent, E = 1: present), S – segmental glomerulosclerosis/adhesion (S0: absent, S1: present), T – the severity of tubular atrophy/interstitial fibrosis (T0 ≤ 25%, T1: 26-50%, T2 > 50%), C – crescents (C0 – absent, C1 < 25% of glomeruli, C2 > 25% of glomeruli) [19]. It is considered that this scale can be used to predict progression of nephropathy in the course of IgAVN as well as correlating with extra-renal symptoms – associated abdominal symptoms predispose to higher MEST-C scores [13]. The presence of crescents in the biopsy samples has an unfavorable prognosis [6, 10, 12, 13, 20].
Since renal biopsy is an invasive procedure, there is a need for less invasive diagnostic procedures. Measurement of biomarkers in urine and in blood seems to be promising method. However, none of them has yet proved to be an established marker of disease and further studies are needed.
In the vast majority of patients with IgAV, because of the self-limited nature of the disease, specific treatment is not required. For IgAVN there are two recommendations that can be used in treatment. The Kidney Disease Improving Global Outcomes (KDIGO) practice guideline on glomerulonephritis provides recommendations for the treatment of IgAVN in children and adults. The second and the newer ones are European consensus-based recommendations for diagnosis and treatment of IgA vasculitis developed by the SHARE initiative (but they are recommended according to the clinical course without results of kidney biopsy) [5].
We are searching for markers that would predict the progress of nephropathy in the course of IgAV or the se- verity of IgAVN.
The aim of the study is to present the currently known biomarkers in IgAV and IgAVN.
Pathogenesis
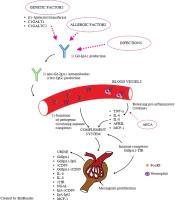
The exact pathogenesis is unknown [21]. It is currently considered that genetic, inflammatory, environmental, and some other factors are significant [22]. According to the latest multi-hit hypothesis [2] four processes are fundamental to the outset and progression of IgAVN: 1) over- production of galactose-deficient IgA1 (Gd-IgA1) and 2) antibodies directed against galactose-deficient IgA1 (anti-Gd-IgA1), 3) formation of pathogenic immune complexes, 4) accumulation of these complexes in the glomeruli [2, 23, 24]. The presence of Gd-IgA1 complexes alone is not pathognomonic for IgAVN. They were confirmed in lupus nephritis, membranous nephropathy, and Sjogren’s syndrome [24]. Furthermore, elevated levels of serum Gd-IgA1 were present among healthy relatives of patients with IgAVN [25]. Therefore, it is considered that its presence in serum does not result in nephropathy development but glomeruli are damaged due to the production of anti- Gd-IgA1 antibodies [26]. Gd-IgA1 components build immune complexes connecting with each other, with IgG antibodies, or other molecules including the Fc fragment of IgA receptor (FcαRI/CD89), and soluble transferrin receptor (sTfR/CD71), increased expression of which is confirmed in IgAVN [2, 11]. Immune complexes consisting of Gd-IgA1 with IgG antibodies are macromolecules and accumulate in the glomerular mesangium, which, through activation of the complement system, leads to inflammation, fibrosis, and ultimately impaired renal function [23, 25]. Complement system activation (via the lectin or alternative pathway) plays a key role in the exacerbation of renal impairment [24-29]. According to the latest findings about IgAVN pathogenesis, abnormality in the function of two receptors in disease development is crucial: FcαRI(CD89) on leucocytes and sTfR (CD71) on mesangial cells of glomeruli [30, 31]. Based on current data, FcαRI/CD89, the specific IgA receptor in the serum of patients with IgAVN, is relevant in the etiology of IgA vasculitis due to the induction of migration of neutrophils and their activation [2]. IgAVN is the inflammatory disease, but the exact role of immune cells is not completely understood. Anti-endothelial cell antibodies (AECA) are significant in this process are released in response to some genetic and/or environmental factors. They cause vascular damage through antibody-dependent cellular cytotoxicity and activation of endothelial cells. At the site of inflammation, mediators are activated, with the release of cytokines and chemokines, e.g. tumor necrosis factor α (TNF-α), interleukin (IL)-6, IL-8, a proliferation-inducing ligand (APRIL), and monocyte chemotactic protein-1 (MCP-1) [28, 32]. The relation between respiratory tract infection and the onset or progression of the disease was confirmed in the scientific literature [33]. Receptor expression is substantial, especially toll-like receptors (TLR), located in mucous membrane tissues, and immune and kidney cells [24]. TLR4 overproduction is associated with nephropathy progression in IgAV. Because impaired glycosylation and galactosylation of IgA and altered gene expression of proteins involved in glycosylation, e.g. β-1,3-galactosyltransferase, core 1 synthase glycoprotein-N-acetylgalactosamine 3-β-galactosyltransferase (C1GALT1) and C1GALT1 specific chaperone 1 (C1GALT1C1), are relevant in disease pathogenesis, they might also influence the disease progression [25].
Genetics
Genetic factors are likely to be crucial in IgAVN pathogenesis. There are some reports of hereditary prevalence or geographical differences in disease occurrence. Also, genetic association studies suggest a contribution of common variants [29]. He et al. in their meta-analysis about IgAV described many different genetic factors predisposing to a severe course of the disease or having a protective effect [34]. Among genes having mutations associated with IgAVN and poor prognosis, angiotensin-converting enzyme (ACE), IL-8, and HLA-B*35 genes were detected although reports from different authors are inconsistent, suggesting the need for further research [34, 35]. The authors highlighted that this disease is not related to a single gene mutation but occurs due to a combination of individual risk factors, which together with environmental factors contribute to the onset of the disease and determine its severity. So far, however, more comprehensive genome-wide association studies (GWAS) have not been published. Two pediatric population studies on IgAN and IgAV with/without nephritis – Genomics of IgA-related disorders in kids, USA (GIGA-kids) and GIGA-Europe Study – are in progress [29].
Biomarkers
Many molecules are involved in the pathogenesis of IgAVN, e.g. biomarkers, which among well-known factors such as proteinuria, arterial hypertension, or lower GFR, have a prognostic role [3, 11, 36].
Biomarkers, by definition, are objectively measurable biological indicators, which suggest the presence of the disease and enable the assessment of its course including its response to treatment [37].
Besides well-known factors such as proteinuria or lower glomerular filtration rate (GFR), the need for the identification of early disease biomarkers was also pointed out [36].
Over the past few years, they have become the subject of many studies on IgAVN, although the obtained results are equivocal and require further investigation, especially among pediatric patients [11].
Gd-IgA1
One of the best known IgAVN biomarkers is Gd-IgA1, i.e. IgA1 with galactose-deficient, O-linked, hinge-region glycans and free N-acetyl galactosamine residue. This is caused by an abnormality of the glycosylating process due to e.g. low expression of GALNT2 (polypeptide N-acetylgalactosaminyltransferase 2), C1GALT1 (core 1 synthase, glycoprotein-N-acetylgalactosamine 3-β-galactosyltransferase 1) and COSMC (core 1 β3-Gal-T-specific molecular chaperone). Gd-IgA1, due to free N-acetyl galactosamine residues, tends to build polymeric structures and stimulate antibody production [38]. Pathogenesis of the abnormal glycosylation of IgA1 is still unknown; however, some suggest exposure to infectious factors (viral or bacterial) and diet in addition to environmental and genetic components.
Many authors have proved the statistically significant higher concentration of this molecule in the serum of patients with IgAVN in comparison to patients with IgAV without nephropathy. A higher concentration of Gd-IgA1 is a risk factor for disease progression and development of kidney failure [11, 23, 24, 29, 36, 39-41]. Suzuki et al. confirmed a higher concentration of Gd-IgA1 among patients with the active form of IgAVN rather than in remission [40]. Gd-IgA1 level might be hereditary since some studies demonstrated its higher concentration levels among healthy relatives of the patients with IgAVN, and galactosylation of IgA1 is partially determined by single nucleotide polymorphism of C1GALT1 [25].
Anti-Gd-IgA1 antibodies
Auto-antibodies, including IgA and IgG class (anti-Gd-IgA1), higher concentrations of which were confirmed among patients with IgAVN rather than with IgAV, are produced as a response against galactose-deficient IgA1 circulating in serum [29, 38]. Furthermore, cross-reactive anti-endothelial cell antibodies (AECA) may emerge as the result of infection with pathogens, which have antigen structures similar to walls of human blood vessels, or under the influence of genetic factors.
Immunoglobulin G (IgG) is of great importance in the progression of IgAV nephropathy. Both Gd-IgA1 level and specific anti-Gd-IgA1 immunoglobulin G may be useful in monitoring the severity of IgAV nephropathy [29, 40].
Gd-IgA1-anti-Gd-IgA1 immune complexes
Circulation and aggregation of immune complexes in serum results in their accumulation in the endothelium of small blood vessels, e.g. in skin, synovial membrane, intestine, and kidneys. Moreover, immune complexes as a result of combining Gd-IgA1 and anti-Gd-IgA1 can activate the component system. They may also stimulate the proliferation of mesangial cells in glomeruli and induce cell proliferation through accumulation in the mesangium. Furthermore, they can release inflammatory mediators and cause kidney failure. Gd-IgA1-anti-Gd-IgA1 immune complexes were detected in the serum of all patients with IgAV independently of renal involvement. Serum levels of these immune complexes are higher among patients with IgAVN in comparison to patients with IgAV without renal involvement [11, 38, 42, 43]. Additionally, sera of patients with IgAV immune complexes do not contain IgG, whereas IgG immune complexes were found in IgAVN patients [2, 43, 44]. Moreover, the concentration of circulating immune complexes correlates with the severity of the disease and its progression [2, 10]. In the study by Pillebout et al. levels of these complexes were only associated with nephritis [11].
The latest identified biomarkers of IgAVN among the adult population include Fc fragment of IgA receptor (FcαRI/CD89) and soluble transferrin receptor (sTfR/CD71).
FcαRI/CD89 is the Fc fragment of the IgA receptor and is present on the surface of phagocytes produced in the bone marrow: neutrophils, eosinophils, and monocytes/macrophages. FcαRI may exist as a trans-membrane receptor on myeloid cells and as a soluble form in the blood (sCD89) [30, 45]. According to Moresco et al., urine sCD89 might be a potential biomarker used to assess IgAVN activity – its levels are decreased among patients with active forms of the disease [46]. Furthermore, the soluble form of the receptor can establish immune complexes with Gd-IgA1 and immunoglobulin G. Complexes are nephrotoxic due to their huge dimensions – they accumulate in glomeruli. Higher levels of them were confirmed in urine samples of patients with IgAVN [10, 11, 25]. The concentration of serum Gd-IgA1-sCD89 complexes is associated with the severity of IgAVN and its progression.
The second significant receptor in the pathogenesis of IgAVN is the soluble transferrin receptor (sTfR/CD71), which might be helpful in the diagnosis and prognosis of IgAVN [41, 47-49]. Serum sTfR of healthy individuals does not penetrate the glomerular membrane. Nevertheless, it may infiltrate into the urine in the case of proteinuria due to damage of the glomerular basement membrane. The study by Delanghe et al. among adult individuals showed that concentration of urinary sTfR correlates with disease severity, i.e. it progresses with proteinuria. Furthermore, a reduction in the amount of the receptor in the urine was observed as a consequence of glucocorticosteroid therapy. It may suggest that the receptor may have various functions – detection of nephropathy in the course of IgAV, monitoring of its progression, or assessment of treatment effectiveness [47]. An and Xia’s study among pediatric patients also confirmed the association of higher TfR levels with an increase in proteinuria [41]. Zhang et al.’s in vitro study proved the impact of Gd-IgA1 on increased expression of soluble transferrin receptor (TfR/CD71), which induces proliferation of mesangial cells [50].
IL-6 and IL-8
Overproduction of the components of protoplasm and chemokines release, e.g. IL-6 and IL-8, is the result of excessive proliferation of mesangial cells.
Interleukin 6 trough T-cells’ activation stimulates mesangial proliferation and fibrosis. IL-8, whose production is stimulated by e.g. IgA1-AECA in walls of small vessels, causes neutrophil chemotaxis, and thus to destruction of vascular endothelial cells [10, 22, 51, 52].
Kimura et al.’s research results demonstrated that serum IL-8 levels among adult patients with IgAVN were significantly higher than in IgAV [53]. However, research by Pillebout et al. and Yuan et al. in children showed elevated serum IL-8 levels in all patients with IgAV regardless of the presence of nephropathy [11, 51], although Yuan et al. detected that its serum levels related to severity of proteinuria in IgAVN [51]. IL-8 urine level among children and adults was significantly higher in a group of patients with IgAVN in comparison to IgAV, which suggests its potential role as a marker of nephropathy progression [11, 36, 51, 52].
Due to its elevated urine concentrations in this group of patients, IL-8 in urine may also be a marker of IgAVN [11, 36].
KIM-1, MCP1, NAG, UAGT and NGAL
The most recent meta-analysis of urine biomarkers in children with IgAV highlights the importance of kidney injury molecule (KIM-1), monocyte chemotactic protein-1 (MCP-1), N-acetyl-β-glucosaminidase (NAG), angiotensinogen (UAGT) and neutrophil gelatinase-associated lipocalin (NGAL) as the most promising molecules in predicting the development of nephritis before the appearance of clinical symptoms [11, 36, 48]. Several studies on the significance of these markers in children with IgAVN have been published in the past few years [32, 54-58]. Marro et al. in their recent study evaluated 124 various urine molecules in children with IgAV, which may be relevant in IgAV/N pathology [54]. Urine UAGT, KIM-1 and MCP-1 levels in children with IgAVN were, among others, statistically significantly higher in comparison to IgAV [54]. These conclusions confirmed the results of other studies, as demonstrated in the meta-analysis by Williams et al. [25, 48], in studies on concentrations of urine MCP-1 [58, 59], and in Yi-fei et al.’s investigation, where higher levels of UAGT in the urine of patients with IgAVN were detected [57]. The results of Dyga et al., in contrast, did not confirm a significant difference between KIM-1 levels in the urine of patients with IgAVN and IgAV [55]. These results need further research.
Vanin-1 is the modern marker of oxidative stress, whose levels are elevated in acute kidney injury (AKI), and might also be a biomarker of the active inflammatory process in children with IgAVN; however, it should be confirmed in a larger group of individuals [60].
Metabolomics
To improve understanding of disease pathogenesis and search for factors modifying its course, some researchers investigate metabolomics, i.e. analysis of metabolism. Demir et al. used metabolic profiling and detected elevated DHAP (18:0), prostaglandin D2/I2, porphobilinogen, 5-methyltetrahydrofolic acid, and N-acetyl-4-O-acetylneuraminic acid/N-acetyl-7-O-acetylneuraminic acid concentrations in patients with IgAVN before nephropathy development in contrast to a group with IgAV but without nephropathy [61]. Sun et al. and their metabolomic analysis provided information that (S)-3-hydroxyisobutyric acid, p-cresol sulfate, and 3-carboxy-4-methyl-5-pentyl-2-furanpropanoic acid are related to progression from IgAV to IgAVN [62]. Moreover, previous studies [61, 63] indicate the influence of platelet-activating factor (PAF), prostaglandins, and arachidonic acid on nephropathy progression.
Summarizing the present evidence, many potential biomarkers for the prediction of nephropathy development in the course of IgAV are currently under investigation (Table 2, Fig. 1). Further studies are important to early planning of treatment and monitoring of its effectiveness, or assessment of disease progression. Nevertheless an increased serum level of Gd-IgA1 is still the most consistent finding in patients with IgAV. For the diagnosis of IgAV with nephritis and to predict prognosis, urinary biomarkers seem to have an advantage over serum biomarkers, because they are more specific to the kidneys. In predicting the development of nephritis in patients with IgAV nephritis before the appearance of clinical symptoms very promising new biomarkers are KIM-1, MCP-1, NAG and UAGT.
Table 2
Analyzed biomarkers in urine and serum
| Biomarker | References | ||
|---|---|---|---|
| Serum | |||
| GdIgA1 | IgAVN > IgAV | [11, 23, 24, 29, 36, 39-41] | |
| Anti-Gd-IgA1 (IgG) | IgAVN > IgAV, related to activity of disease in patients with IgAVN | [30, 38, 40] | |
| Immune complexes containing GdIgA1 | High level of IgG-GdIgA1 (large molecules) only in IgAVN | [2, 42] | |
| Non-IgG complexes (small molecules) IgAVN > IgAV | [38, 43] | ||
| NGAL | Present in IgAVN and IgAV, unrelated to proteinuria and GFR | [11, 36] | |
| IL-8 | IgAVN > IgAV | [53] | |
| IgAV > healthy control | [11, 51] | ||
| In IgAVN related to severity of proteinuria | [51] | ||
| Urine | |||
| GdIgA1 | IgAVN > IgAV | [11, 36, 44] | |
| GdIgA1-IgG | IgAVN > IgAV | [11, 29] | |
| sCD89 | Active IgAVN < non-active IgAVN | [40] | |
| GdIgA1-sCD89 | IgAVN > IgAV | [10, 11, 25] | |
| IL-6 | IgAVN > IgAV | [11, 36] | |
| IL-8 | IgAVN > IgAV | [11, 36, 51, 52] | |
| sTfR | IgAVN > IgAV | [41, 48] | |
| NGAL | Present in IgAV and IgAVN, unrelated to proteinuria and GFR | [11, 36] | |
| IgAVN > IgAV | [29, 36] | ||
| IgA-sCD89 | Elevated only in IgAVN | [11, 36] | |
| IgA-IgG | Elevated only in IgAVN | [11, 36] | |
| MCP-1 | IgAVN > IgAV | [48, 59] | |
| Vanin-1 | Active IgAVN | [60] | |
[i] Gd-IgA1 – galactose-deficient IgA1, anti-Gd-IgA1 (IgG) – antibodies directed against galactose-deficient IgA1 (class IgG), NGAL – neutrophil gelatinase-associated lipocalin, GdIgA1-IgG – complexes consisted of GdIgA1 and IgG, sCD89 – soluble Fc fragment of IgA receptor, GdIgA1-sCD89 – complexes consisting of GdIgA1 and sCD89, IL-6 – interleukin 6, IL-8 – interleukin 8, sTfR – soluble transferrin receptor, IgA-sCD89 – complexes consisting of IgA and sCD89, IgA-IgG – complexes consisting of IgA and IgG, MCP-1 – monocyte chemoattractant protein 1
Fig. 1
Pathogenesis and some biomarkers in immunoglobulin A (IgA) vasculitis (IgAV) and IgA vasculitis with nephritis (IgAVN)
C1GALT1 – core 1 synthase glycoprotein-N-acetylgalactosamine 3-β-galactosyltransferase, C1GALTC2 – C1GALT1 specific chaperone 1, Gd-IgA1 – galactosedeficient IgA1, anti-Gd-IgA1 (IgG) – antibodies directed against galactose-deficient IgA1 (class IgG), TNF-α – tumor necrosis factor α, IL-6 – interleukin 6, IL-8 – interleukin 8, APRIL – a proliferation-inducing ligand, AECA – anti-endothelial cell antibodies, MCP1 – monocyte chemoattractant protein 1, sCD89 – soluble Fc fragment of IgA receptor, GdIgA1-sCD89 – complexes consisted of GdIgA1 and sCD89, sTfR – soluble transferrin receptor, NGAL – neutrophil gelatinase- associated lipocalin, IgA-sCD89 – complexes consisted of IgA and sCD89, GdIgA1-IgG – complexes consisting of GdIgA1 and IgG, IgA-IgG – complexes consisting of IgA and IgG