Introduction
Ulcerative colitis (UC) is a persistent inflammatory disease of the colon, with high treatment costs and increasing global incidence and prevalence [1-3]. Although UC is predominantly found in Western nations, its prevalence has been rising in recently industrialized regions, such as those in Asia, as societies adopt more Western lifestyles [4, 5]. The pathogenesis of UC is influenced by various factors, including dysregulated immune responses, microbial imbalances, genetic susceptibility, and environmental factors [6-9]. Nevertheless, the specific initiating and compounding factors of inflammation remain unclear [10]. Research to date has concentrated on the disruption of immune mechanisms, both adaptive and innate [11-13]. γδ T cells, which are present in both tissues and blood, are unconventional T lymphocytes that are identified by a restricted repertoire of T cell receptors (TCRs). The major histocompatibility complex does not limit the activity of γδ T cells, and they do not rely on the presentation and processing of antigens, as opposed to αβ T cells. This enables them to serve as an initial defensive barrier during inflammation that is both sterile and non-sterile [14]. In addition to their involvement in immune modulation, tumor monitoring, and the repair of intestinal epithelial injury, γδ T cells are linked to the onset of other immunological-mediated illnesses, such as inflammatory bowel disease (IBD) [15-17]. Recent advancements in the field of IBD have revealed the intriguing role of a novel subset of γδ T cells, distinguished by the co-expression of TCRγδ and CD8. This subset, known as CD8+ γδ T cells, is evident in both the intestinal mucosa (IM) and the peripheral blood (PB), with notable enrichment in the former [18]. As demonstrated in previous studies, these cells, similar to γδ T cells, are capable of detecting diverse microbes along with compromised or altered cells of the host organism. Their cytotoxic activity encompasses both secretory and non-secretory mechanisms. These mechanisms involve the release of perforins and granzymes, in addition to the interactions with Fas and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) [15, 18-21]. According to one study, CD8+ γδ T cells represented one of the least abundant subsets observed in CD patients with surgical relapse. In addition, the lowest level of CD8+ γδ T cells was also observed in the fistulizing phenotype. CD8+ γδ T cells are closely related to the disease progression of CD patients [22]. Our previous study explored CD8+ γδ T cell immunological features in Crohn’s disease patients [23]. However, CD8+ γδ T cells’ specific role in UC is not entirely elucidated. To address this gap, our investigation focused on examining the immunological properties of UC patients’ peripheral and intestinal CD8+ γδ T cells and correlating these properties with disease activity (DA). Our findings provide potentially illuminating new avenues for understanding the immunopathogenesis of UC and identifying novel therapeutic targets.
Material and methods
Cohort characteristics
Fresh PB specimens were obtained from two cohorts: healthy controls (HCs; n = 38) and individuals with UC (n = 30). Intestinal biopsy samples were collected from two cohorts: HCs (n = 21) and individuals with UC (n = 21). Individuals with UC were diagnosed based on clinical assessment, radiological imaging, histological examination, and endoscopy. All participants were screened to exclude other autoimmune disorders, cancers, and communicable illnesses such as hepatitis B or tuberculosis. No immunosuppressants, bowel surgery, organ/marrow transplants, or blood transfusion had been administered to them in the last year. Patients who were in remission were omitted from the study, which only included those who were in the active phase. Disease severity was evaluated using the Ulcerative Colitis Disease Activity Index (UCDAI), with values ≤ 2 signifying remission, 3-5 mild activity, 6-10 moderate activity, and 11-12 severe activity. PB samples from patients with UC were categorized as exhibiting moderate (n = 17) or severe (n = 13) DA. Similarly, intestinal biopsy samples were categorized as exhibiting moderate (n = 14) or severe (n = 7) DA. Fresh human blood and intestinal biopsy specimens necessary for the investigation were procured from the Department of Gastroenterology at Zhejiang University School of Medicine’s Sir Run Run Shaw Hospital, located in Hangzhou, China. The research received ethical approval from the Sir Run Run Shaw Hospital Ethics Committee (number: 2024-0466) and was performed in accordance with the Declaration of Helsinki principles. Written informed consent was given by everyone who participated in the study. No substantial disparities in age or gender distribution were observed among the study cohorts. Tables 1 and 2 outline the clinical features of the sample donors.
Table 1
Clinical features of peripheral blood (PB) donors
| Cohort | HC | UC |
|---|---|---|
| Cases | 38 | 30 |
| Sex (male/female) | 20/18 | 18/12 |
| Age (years) | 33.5 (21-50) | 44.5 (15-65) |
| UCDAI score | ND | 10 (6-12) |
| Disease activity (mild/moderate/severe) | ND | 0/17/13 |
Sample acquisition and processing
All participants had a 5 ml sterile blood sample drawn and collected using EDTA-anticoagulated tubes. Whole blood specimens were processed via Ficoll density gradient centrifugation for the extraction of mononuclear cells from PB. Mucosal tissues from the intestines of UC patients and HCs were collected during endoscopic procedures and immediately stored in sterile PBS for transport to the laboratory. After mincing the tissues, they were placed in Hank’s Balanced Salt Solution with EDTA and DTT for 40 minutes. Subsequently, they were subjected to enzymatic digestion at 37°C for 2 hours with collagenase type IV and DNase. Thereafter, a nylon mesh (70-µm pore size) was used to filter the cell suspension that was obtained before being rinsed twice with sterile PBS.
Fluorescence-activated flow cytometry analysis
Samples from both circulating blood and gut mucosa were employed to evaluate the characteristics and functions of CD8+ γδ T lymphocytes. These specimens underwent exterior labeling, then fixation and permeabilization for internal staining. The following fluorochrome-linked antibodies were used: Alexa Fluor 700-bound anti-human gran- zyme B recombinant mAb, PE-bound anti-human PD-1 mAb, Zombie Red Viability Dye FITC-bound anti-human CD3 mAb, APC/Fire 750-bound anti-human CD8 mAb, Brilliant Violet 510-bound anti-human CD45 mAb, Brilliant Violet 421-bound anti-human TCR γ/δ mAb, PE-bound anti-human perforin mAb, PE/Cyanine7-bound anti-human TRAIL mAb, and Brilliant Violet 785-bound anti-human HLA-DR mAb, all from BioLegend, USA. Subsequently, the DxFLEX Flow Cytometer (Beckman, USA) was employed to quantify the intensity of fluorescence, capturing no less than 100,000 events per specimen. Data interpretation was conducted using the FlowJo program (Tree Star). Figure 1A shows the gating method. The gating strategy of CD8+ γδ T is CD3+ TCR γδ+ CD8+. Subsequently, fluorescence minus one (FMO) controls were employed to decipher the persisting patterns of fluorescent marker expression (perforin, HLA-DR, granzyme B, PD-1, and TRAIL), with the FMO outcomes presented in the supplementary material (Suppl. Fig. 1).
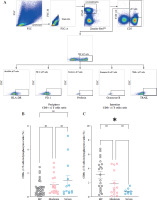
Fig. 1
Levels of intestinal CD8+ γδ T cells in individuals with ulcerative colitis (UC) decrease with increasing disease activity (DA). A) Complete gating strategy for flow cytometry used with the study participants. B) Aggregated data on the proportion of CD8+ γδ T cells (CD3+ TCR γδ+ CD8+) in the PB across HCs, individuals with moderate UC activity, and individuals with severe UC activity. C) Aggregated data on the proportion of CD8+ γδ T cells (CD3+ TCR γδ+ CD8+) in the IM among healthy controls (HCs), individuals with moderate UC activity, and individuals with severe UC activity. Results are shown as mean ± SEM. Statistically significant results are show as follows: *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001
Statistical analysis
The statistical data analysis was conducted using GraphPad Prism (v.9.4.0, San Diego, CA, USA) software. Comparative analyses were performed with Mann-Whitney U tests for two groups and two-way ANOVAs for many subgroups. Except where otherwise specified, data are presented as means, and graphical data are represented as the mean ± SEM. Two-sided p values below 0.05 were computed to establish statistical significance.
Results
The level of intestinal CD8+ γδ T cells in UC decreases with increasing DA
We analyzed the proportion of CD8+ γδ T cells in the PB and IM of all the participants using flow cytometry. Our findings revealed no notable variance in the levels of peripheral CD8+ γδ T cells among the healthy cohort, UC individuals with moderate DA cohort, and UC individuals with severe DA cohort. We found that the level of IM CD8+ γδ T cells in individuals with moderate UC activity remained relatively stable compared to HCs. However, it significantly decreased in patients with severe UC activity (Fig. 1B, C). This indicates that when DA increases in UC patients, the proportion of IM CD8+ γδ T cells decreases.
Intestinal CD8+ γδ T cell immune status shifts from activation to suppression with increased DA in individuals with UC
The presence of the activation indicator HLA-DR as well as the inhibitory factor PD-1 was measured to evaluate the immunological state of CD8+ γδ T cells in UC patients displaying differing levels of DA. Our analysis revealed no notable variations in HLA-DR manifestation on peripheral CD8+ γδ T cells among three cohorts (Fig. 2A). In individuals suffering from moderate UC activity, significant upregulation of HLA-DR on intestinal CD8+ γδ T cells was observed compared with HCs, indicating enhanced immune cell activation.
Fig. 2
Intestinal CD8+ γδ T cell immunological status shifts from activation to suppression with increased disease activity (DA) in individuals with ulcerative colitis (UC). A) Data comparing CD8+ γδ T cell expression of HLA-DR in peripheral blood (PB) among healthy controls (HCs), individuals with moderate UC activity, and individuals with severe UC activity. B) Data comparing the CD8+ γδ T cell expression of PD-1 in PB among HCs, individuals with moderate UC activity, and individuals with severe UC activity. C) Data comparing CD8+ γδ T cell expression of HLA-DR in the IM among HCs, individuals with moderate UC activity, and individuals with severe UC activity. D) Data comparing CD8+ γδ T cell expression of PD-1 in the IM among HCs, individuals with moderate UC activity, and individuals with severe UC activity. HLA-DR – activation marker, PD-1 – immunosuppressive molecule. Results are shown as mean ± SEM. Statistically significant results are shown as follows: *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001
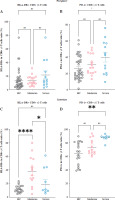
However, in patients with severe UC activity, this increase was absent, with HLA-DR expression on these cells markedly diminished relative to the individuals with moderate UC activity (Fig. 2C). Conversely, PD-1 levels on both peripheral and intestinal CD8+ γδ T lymphocytes were significantly elevated in individuals with severe UC activity, with no significant changes observed in individuals with moderate UC activity (Fig. 2B, D). According to the results, intestinal CD8+ γδ T lymphocytes in patients with moderate UC activity exhibit immunoactivation, whereas those with severe UC activity display immunosuppression. This suggests a shift in intestinal CD8+ γδ T cells’ immunological status from activation to suppression as the disease progresses, potentially impacting their cytotoxic function.
Intestinal CD8+ γδ T cell cytotoxicity is impaired as DA increases in individuals with UC
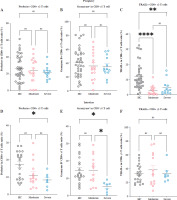
To better understand the CD8+ γδ T cells’ cytotoxic capabilities in UC individuals exhibiting differing levels of DA, we examined the levels of cytotoxic molecules – TRAIL, granzyme B, and perforin – in both PB and IM CD8+ γδ T cells. In PB CD8+ γδ T cells, we detected no significant variations in the proportions of perforin and granzyme B across the three cohorts. However, compared to healthy controls, UC patients recorded a considerable decline in TRAIL levels in peripheral CD8+ γδ T cells (Fig. 3A-C). In the intestinal compartment, no notable disparities were detected regarding how these molecules are expressed in CD8+ γδ T cells in those with moderate UC activity and HCs. However, in patients with severe UC activity, there was a notable decline in the proportions of granzyme B and perforin in CD8+ γδ T cells, while TRAIL expression remained unchanged (Fig. 3D-F). These findings indicate that while the cytotoxic capabilities of IM CD8+ γδ T cells remain largely intact in those with moderate UC activity, they are significantly diminished in individuals with severe UC. This implies that the impairment in cytotoxic function is associated with the stage of the disease.
Fig. 3
Intestinal CD8+ γδ T cell cytotoxicity is impaired as disease activity (DA) increases in individuals with ulcerative colitis (UC). A-C) Levels of cytotoxic molecules – perforin, granzyme B, and TRAIL – in CD8+ γδ T cells of peripheral blood (PB) samples from healthy controls (HCs), individuals with moderate UC activity, and individuals with severe UC activity are demonstrated by the pooled data. D-F) Levels of cytotoxic molecules – perforin, granzyme B, and TRAIL – in CD8+ γδ T cells of the intestinal mucosa (IM) samples from HCs, individuals with moderate UC activity, and individuals with severe UC activity are demonstrated by the pooled data. Perforin, granzyme B, and TRAIL – cytotoxic molecules. Results are shown as mean ± SEM. Statistically significant results are shown as follows: *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001
Evaluation of CD8+ γδ T cells using ROC curves in UC
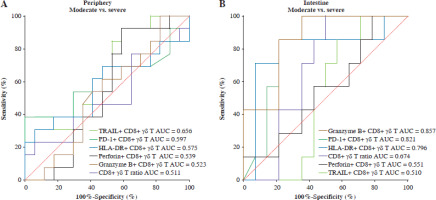
Our examination of the immunological features exhibited by CD8+ γδ T cells across various stages of UC DA revealed distinct profiles. As demonstrated by the ROC curve, the intestinal granzyme B+ CD8+ γδ T cell ratio (AUC = 0.857), intestinal PD-1+ CD8+ γδ T cell ratio (AUC = 0.821), and intestinal HLA-DR+ CD8+ γδ T cell ratio (AUC = 0.796) can serve as robust biomarkers for differentiating between patients with moderate and severe UC activity (Fig. 4B). These markers demonstrated high specificity (64.29%, 64.29%, and 92.86%) and sensitivity (100%, 100%, and 71.43%), respectively. As detailed in Table 2, additional parameters did not exhibit statistical significance.
Fig. 4
Assessment of the ROC curve for CD8+ γδ T cells in individuals with ulcerative colitis (UC). A) Differentiating between individuals with moderate UC activity and those with severe UC activity using ROC curve analysis of biomarkers in CD8+ γδ T cells from peripheral blood (PB). B) Differentiating between individuals with moderate UC activity and those with severe UC activity using ROC curve analysis of biomarkers in CD8+ γδ T cells from IM. AUC – area under the curve
Discussion
Ulcerative colitis is widely recognized as a persistent inflammatory disease predominantly affecting the colon, with a documented increase in both incidence and prevalence globally [3]. Historically, research into the etiology of UC has focused on aberrations in immune system responses, particularly emphasizing traditional T cells [13]. However, in recent years, a paradigm shift has occurred, with emerging evidence suggesting that atypical γδ T cells likewise perform a critical function in the onset and advancement of IBD [24]. Studies have shown that γδ T cells have the function of tissue healing, and their deficiency can lead to the decrease of the healing ability of ulcers as well as fistulas in CD patients [25, 26]. Notably, a subgroup of γδ T cells distinguishable by expressing both TCRγδ and CD8 has been identified in patients with IBD and is denoted as CD8+ γδ T cells. Notwithstanding these findings, the precise significance of CD8+ γδ T cells in the development of UC has not been completely clarified [18]. Our investigation focused on the immunological attributes of CD8+ γδ T cells in both the PB and the IM of UC patients, determining their association with DA.
Our investigation involved a comprehensive examination of CD8+ γδ T cells in the IM and PB of individuals with UC across differing levels of DA. Initially, we found that while the proportions of PB CD8+ γδ T cells remained relatively constant, a considerable decline in the proportion of IM CD8+ γδ T cells was associated with increased severity of disease. In previous animal models of different types of IBD, it was also observed that a deficiency of γδ T cells led to aggravation of intestinal inflammation and higher mortality [27-29]. We extend these findings by demonstrating that CD8+ γδ T cells can similarly contribute to intestinal homeostasis.
Our investigation revealed a substantial decline in the levels of IM CD8+ γδ T cells in individuals with UC under conditions of heightened inflammation. This observation led us to investigate the mechanism by which the immune status and function of these cells are influenced by the inflammatory milieu. Subsequent research unveiled that IM CD8+ γδ T cells in individuals with moderate UC activity highly expressed HLA-DR, which indicated that they were in an immune activation state. Conversely, both PB and IM CD8+ γδ T cells in individuals with severe UC activity highly expressed PD-1, which indicated that they were both in immunosuppression. This dichotomy suggests that as DA progresses, CD8+ γδ T cell immunological status in UC patients may transition from activation to immunosuppression, potentially leading to cytotoxic damage. It is well documented that anti-inflammatory properties may be exerted by CD8+ γδ T cells by removing infected cells, tumor cells, or cells stressed by inflammation [18]. These cells can directly exert cytotoxicity through both secretory (e.g., granzymes and perforins) and non-secretory (e.g., Fas and TRAIL) pathways [15, 18-21]. To investigate the CD8+ γδ T cell cytotoxicity further, we assessed the levels of critical cytotoxic molecules – granzyme B, TRAIL, and perforin – in these cells across different stages of UC DA. Our analysis revealed that the levels of granzyme B and perforin in peripheral CD8+ γδ T cells remained similar between individuals with moderate and severe DA. This suggests that these secretory pathways may not be the primary factors contributing to the observed phenotypic changes. In contrast, UC patients exhibited a substantial decline in TRAIL expression in the peripheral CD8+ γδ T cells, indicating potential attenuation of the non-secretory apoptotic pathway. Furthermore, the IM CD8+ γδ T cell cytotoxicity in patients with moderate UC activity remained unaltered and did not exhibit enhancement with increased cellular activation. However, in individuals with severe UC activity, the cytotoxicity of these cells in the intestines was markedly diminished. We believe that this might be related to the immunosuppression caused by the high expression of PD-1 in individuals with severe UC activity.
These findings underscore the dynamic interplay between DA and the immunological activity of CD8+ γδ T cells, suggesting a possible shift of IM CD8+ γδ T cells from immune activation to immune suppression as the disease progresses.
The progression from immune activation to suppression and the concomitant impairment of cytotoxicity in IM CD8+ γδ T cells as DA increases may have significant implications for the pathophysiology of UC. Importantly, our study identified that the proportions of intestinal granzyme B+ CD8+ γδ T cells, intestinal PD-1+ CD8+ γδ T cells, and intestinal HLA-DR+ CD8+ γδ T cells serve as discriminative biomarkers for disease severity, exhibiting high specificity and sensitivity. These biomarkers could provide a valuable diagnostic and prognostic framework for distinguishing between moderate and severe cases of UC.
In conclusion, our findings delineate the immunological landscape of CD8+ γδ T cells in UC, underscoring their promise as therapeutic targets and diagnostic indicators. The observed reduction in IM CD8+ γδ T cells concurrent with heightened DA, the transition from an activated to a suppressed immune status, and the accompanying decline in cytotoxicity all provide a foundation for further investigation into the immunotherapeutic modulation of UC.

