Introduction
The atopic march, a well-recognized paradigm in allergic diseases, describes the natural progression from early-onset food allergies and atopic dermatitis in infancy to allergic rhinitis and asthma in childhood and beyond. This sequential manifestation underscores the interconnectedness of allergic conditions and their significant impact on pediatric health [1]. Food allergy, particularly cow’s milk allergy (CMA), often represents the initial allergic sentinel, affecting approximately 2-3% of infants in developed countries and frequently preceding the development of respiratory allergies [2]. Indeed, immunoglobulin E (IgE)-mediated CMA has been strongly implicated in the subsequent development of asthma, highlighting the systemic nature of allergic sensitization.
Bronchial asthma represents the most common chronic respiratory disease worldwide, significantly affecting the pediatric population, particularly in economically developed countries [3]. The persistent increase in asthma prevalence in many European countries, as documented by epidemiological studies, is alarming. For instance, data from repeated surveys from Poland indicate a significant rise in asthma prevalence from 3.4% in 1993 to 12.6% in 2014, confirming an exponential trend over two decades [4]. This dynamic increase highlights the complex etiopathogenesis of the disease, where environmental factors, genetic predisposition, and lifestyle influences intertwine. Notably, the T2 endotype represents the most prevalent form of asthma, particularly in children, accounting for over 90% of asthma cases [5]. T2 inflammation profile is marked by the production of key cytokines including interleukin (IL)-4, IL-5, and IL-13. IL-4 promotes IgE class switching in B cells, leading to IgE production [6]. While inhaled corticosteroids (ICS) form the cornerstone of asthma management, a subset of patients, particularly those with early-onset, severe allergic asthma, remain poorly controlled despite high-dose conventional therapies, including systemic corticosteroids. These patients experience frequent exacerbations, hospitalizations, and a significantly impaired quality of life, representing a considerable clinical challenge.
The complexity is further amplified in patients with allergic multimorbidity, where co-existing conditions such as food allergy and asthma exacerbate disease burden and management difficulty. In this context, targeted biological therapies offer promising avenues. This case report details the clinical course of a 7-year-old child with a history of severe, early-onset CMA complicated by poorly controlled, severe persistent asthma. We report the dual effect of the anti-IgE monoclonal antibody omalizumab in improving both asthma control and tolerance to high-risk foods, thereby highlighting its potential in managing pediatric allergic multimorbidity.
Case description
The patient, a 7-year-old boy, presented with a complex history of allergic disease initiated in early infancy. A significant family history of allergy was noted, with his mother being diagnosed with allergic conditions, placing him at an increased risk of developing allergies (estimated 20-40%) [7]. His perinatal history was unremarkable, and he was breastfed for the first four months of life. However, introduction of cow’s milk-based formula triggered immediate gastrointestinal symptoms including vomiting within 30 minutes of ingestion, colic, and constipation. Following this reaction, he was transitioned to an extensively hydrolyzed formula which was well tolerated, indicative of early-onset CMA. Furthermore, the patient had been maintained on a strict total milk-free diet since the age of 4 months due to his documented CMA, leading to the development of milk aversion. His persistent allergic burden significantly impacted his quality of life. Surprisingly, he did not develop significant atopic eczema in infancy.
His early childhood was further complicated by recurrent episodes of virus-induced wheezing requiring repeated hospitalizations and treatment with ICS and short-acting beta-agonists. These recurrent respiratory issues evolved into severe persistent bronchial asthma, required GINA step 4 treatment, which proved to be poorly controlled despite adherence to guideline-recommended therapy. His asthma regimen included a medium-high dose combination of fluticasone/formoterol, a leukotriene receptor antagonist, and anti-allergic medications. He demonstrated good adherence to his medication regimen and appropriate inhaler technique. Despite this comprehensive approach, he continued to experience frequent asthma exacerbations (3 asthma flares) requiring admissions to hospital, including systemic corticosteroid administration during 2023. The asthma symptoms of our patient were characterized by numerous attacks of wheezing, particularly prominent at night. The presence of distant wheezing indicated significant airway obstruction. In addition to asthma, the patient was diagnosed with mild laryngopharyngeal reflux disease, which is clinically relevant as it may contribute to the observed asthma severity. He exhibited reduced lung function and marked bronchial hyperresponsiveness. Exercise tolerance was limited, sleep quality was impaired, and respiratory infections were often protracted. His Asthma Control Test (ACT) scores consistently ranged between 13 and 16, indicating unsatisfactory asthma control. Elevated fractional exhaled nitric oxide (FeNO) levels, ranging from 45 to 110 ppb, further indicated persistent airway inflammation and an increased risk of asthma exacerbations. Of particular note, the patient experienced two episodes of COVID-19 during the period of 2022-2023, both with flu-like disease courses. He was unvaccinated against COVID-19. As highlighted by research on post-COVID-19 pulmonary sequelae in children by Özdemir, even mild COVID-19 can contribute to persistent respiratory symptoms and functional abnormalities [8].
Laboratory investigations revealed marked allergic sensitization to timothy grass (Phl p 1) and cat dander (Fel d 1), consistent with his clinical history of seasonal rhinoconjunctivitis during summer grass pollen exposure and eosinophilic inflammation. Total serum IgE levels were significantly elevated and demonstrated a rising trend over time: initially measured at 289 IU/l in November 2020, increasing to 737 IU/l in July 2021, and further fluctuating but reaching a peak of 1690 IU/l in October 2024. Eosinophil cationic protein ECP, a marker of eosinophil activity, was markedly elevated, exceeding 200 µg/l (normal range: < 18.8 µg/l), indicative of significant eosinophilic inflammation. Specific IgE testing confirmed strong sensitization to cow’s milk proteins. Specific IgE to extracts was elevated in November 2022. Component-resolved diagnostics using the multiplex ImmunoCAP ISAC test (Thermo Fisher Scientific Inc., Sweden) in March 2023 and Phadia monocomponent assays in January and August 2024 revealed persistently high levels of specific IgE to key cow’s milk allergens: alpha-lactalbumin (Bos d 4), β-lactoglobulin (Bos d 5), and thermostable casein (Bos d 8) (Table 1). Clinically significant elevated specific IgE levels, notably against the Bos d 8 component, strongly pointed towards severe, ongoing CMA and a high likelihood of anaphylaxis upon even minimal milk ingestion.
Table 1
Immunoglobulin E (IgE) levels to cow’s milk allergens over time
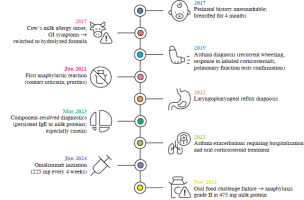
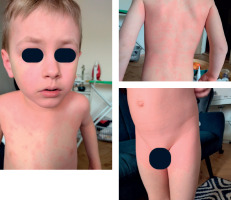
This clinical suspicion was corroborated by a history of multiple documented reactions following cow’s milk exposure (Fig. 1). In June 2021, at the age of 4 years, the patient experienced an anaphylactoid reaction triggered by contact with milk, manifesting as contact urticaria and pruritus. Subsequently, in March 2022, he suffered anaphylaxis after consuming potatoes prepared with butter, exhibiting dyspnea, suffocation, cough, distant wheezing, tachycardia, and urticaria. A further anaphylactic episode occurred in November 2022, without any identifiable dietary error, characterized by nasal congestion, dyspnea, cough, distant wheezing, vomiting, and urticaria. In January and May 2023, anaphylactic reactions were triggered by inhalation exposure in the kitchen during cooking with grilled cheese, presenting with nasal congestion, dyspnea, cough, and facial edema. These repeated anaphylactic reactions, triggered by various routes of exposure, underscored the severity and persistent nature of his CMA. The patient’s anaphylactic reactions consistently presented with mucocutaneous and respiratory symptomatology. These episodes were classified as Grade II anaphylaxis according to symptom severity and system involvement, requiring epinephrine administration as per established anaphylaxis management protocols [9]. Quality of life assessment using the Food Allergy Quality of Life Questionnaire – Parent Form (FAQLQ-PF) revealed a score of 36 points (maximum 76 points), indicating that the patient’s quality of life was significantly impacted by his food allergy [10]. Specifically, parental responses highlighted that the child “quite significantly” experienced anxiety about accidental consumption of risky foods, felt different from his peers, and experienced emotional distress due to his allergy. Social limitations due to the need to find safe restaurants and holiday destinations were also reported as “quite significant”. Furthermore, the parents reported that the child was “mildly” frustrated by dietary restrictions and that both the child and parents experienced “extremely significant” fear and anxiety related to trying new foods and the potential for allergic reactions.
Fig. 1
Clinical manifestation of anaphylaxis with predominant mucocutaneous presentation (published with the consent of the legal guardian)
Given the patient’s persistent severe allergic asthma, early-onset disease, sensitization to perennial aeroallergens, history of more than two severe exacerbations in the preceding year, and elevated total IgE levels within the recommended dosing range, omalizumab therapy was initiated in June 2024. Consistent with established dosing guidelines, the omalizumab dose for this 7-year-old patient was calculated based on his weight (21 kg) and baseline total serum IgE level (403 IU/l) at the time of treatment initiation [11]. Biologic therapy was administered subcutaneously every 4 weeks at a dose of 225 mg, divided into 150 mg and 75 mg syringes. Clinical validation studies show that this regimen achieves > 95% target engagement (free IgE < 50 ng/ml) in 92% of pediatric patients [8]. The rationale for omalizumab therapy was to target the underlying IgE-mediated inflammation driving both his severe allergic asthma and his persistent CMA, aiming to improve asthma control, reduce exacerbation frequency, and potentially increase his tolerance threshold to cow’s milk.
Following the initiation of omalizumab therapy, a notable positive trend was observed in the patient’s clinical status, particularly within the first two months after the first two doses of omalizumab administration. Improvement in asthma control was reported by the patient and his parents. He experienced a significant decrease in the frequency and severity of asthma symptoms, with reduced nighttime wheezing and improved exercise tolerance. Importantly, during this initial period after omalizumab initiation, the patient experienced no asthma exacerbations requiring hospital admission or systemic corticosteroids. Objectively, improvements in pulmonary function parameters were noted during follow-up visits. While detailed spirometry data require further evaluation over a longer period, initial assessments indicated a trend towards increased FEV1 (+12%) and improved peak expiratory flow rates (+25%). Furthermore, repeat FeNO measurements demonstrated a decrease from pre-treatment levels (20-22 ppb), although still slightly elevated, suggesting a reduction in airway inflammation. ACT scores also showed a clinically significant improvement, consistently ranging between 21 and 23 points, indicating much better asthma control compared to pre-treatment scores. Given the marked improvement in asthma control with omalizumab, we plan to initiate allergen immunotherapy targeting causal grass allergens, supported by evidence of its efficacy in asthma treatment [12].
Following the promising initial improvements in asthma control and pulmonary function after two doses of omalizumab, further evaluation focused on assessing the impact of therapy on the patient’s CMA. After five months of treatment, corresponding to five doses of omalizumab administration, an open-label oral food challenge with fresh cow’s milk was conducted. This procedure was performed in accordance with the established guidelines of the Czech Society of Clinical Immunology and Allergy, using an IgE-mediated CMA test protocol. The cumulative target dose for the challenge was 144.4 ml of fresh cow’s milk, representing a total of 4765 mg of milk proteins [13]. The patient unfortunately experienced an anaphylactic reaction during the food challenge. The reaction occurred after the fifth administered dose of cow’s milk, which was 10 ml (330 mg of protein, cumulative dose 475 mg of protein, representing approximately 10% of the total planned dose). The anaphylactic reaction was classified as grade II, characterized by facial erythema and edema, generalized pruritus, nasal congestion, and cough. In response to this reaction, standard anaphylaxis management protocols were immediately implemented, including the administration of intramuscular epinephrine and other appropriate supportive medications to manage the acute symptoms. Figure 2 summarizes the patient’s timeline from allergy onset to omalizumab therapy and subsequent food challenge.
Discussion
Omalizumab, the biologic agent employed in this case, is a humanized anti-IgE monoclonal antibody strategically designed to target the IgE-mediated inflammatory pathway central to allergic diseases. This innovative therapy, first approved by the U.S. Food and Drug Administration (FDA) in 2003 and subsequently by the European Medicines Agency (EMA) in 2005, has been available on the market for nearly two decades [14]. Its therapeutic action stems from several key biological mechanisms. Primarily, omalizumab binds to the Fc fragment of free IgE in the serum, effectively reducing circulating IgE levels to < 5% of baseline values within 24 hours of administration by forming biologically inert complexes [15]. Furthermore, it inhibits the binding of IgE to the high-affinity FcεRI receptor on effector cells such as mast cells and basophils and downregulates FcεRI expression on these cells. This dual action leads to the stabilization of mast cells, raising their threshold for activation and degranulation, thereby reducing the release of pro-inflammatory mediators [16, 17]. Ultimately, omalizumab diminishes the activation and infiltration of eosinophils into target tissues, thus directly addressing the key inflammatory processes characteristic of T2-high allergic conditions such as severe asthma [18].
Clinical trials demonstrate that omalizumab reduces annualized asthma exacerbation rates by 50-75% in pediatric populations while significantly improving lung function parameters and quality of life metrics [19, 20]. Children receiving omalizumab show improved asthma control, with higher ACT scores and better lung function indicators such as FEV1% [21]. Omalizumab also reduces hospitalization rates and allows for a reduction in inhaled corticosteroid doses or exclusion from oral corticosteroid therapy [22, 23]. Long-term safety data from post-marketing surveillance studies confirm the drug’s favorable risk-benefit profile, with injection-site reactions being the most frequently reported adverse event [22, 24].
Beyond its established role in asthma management, omalizumab has emerged as a promising therapeutic strategy for pediatric food allergies, a condition that poses significant management challenges for clinicians and families alike. The stringent allergen avoidance diets and constant preparedness for emergency interventions associated with severe food allergies significantly impact quality of life. However, recent advancements in immunomodulatory therapies, particularly omalizumab, offer groundbreaking approaches to mitigate allergic reactions. The proposed mechanism of action of omalizumab effectively raises the threshold for allergen-induced activation, creating a crucial “buffer” against accidental exposures and reducing the risk of severe reactions [25]. Importantly, while omalizumab provides this temporary immunomodulation, it does not eliminate the underlying allergy, necessitating ongoing administration to sustain its benefits [26]. This dependency on continuous therapy is underscored by evidence demonstrating that treatment withdrawal may precipitate a rapid decline in clinical benefits, including resurgence of asthma symptoms and heightened risk of severe allergic reactions [25, 27]. The clinical efficacy of omalizumab in food allergy is powerfully illustrated by pivotal trials such as the OUTMATCH study. This phase 3 trial demonstrated omalizumab’s capacity to significantly increase reaction thresholds in children with multiple food allergies. After 16-20 weeks of treatment, a remarkable 67% of participants treated with omalizumab tolerated at least 600 mg of peanut protein, compared to a mere 7% in the placebo group. Comparable and significant improvements were observed for other common allergens such as cashew, milk (66% of children were able to tolerate 1000 mg of milk proteins vs. 11% in the placebo group), and egg, with many children achieving tolerance to doses exceeding those encountered in typical accidental exposures [28]. These findings are consistent with smaller randomized controlled trials demonstrating a significant 2- to 488-fold increase in tolerance thresholds for milk, egg, peanut, and fish allergens during omalizumab therapy [29, 30]. Furthermore, longitudinal observational studies have reported sustained benefits of omalizumab treatment, extending to 12 months and beyond. These studies show improvements in food-related quality of life scores and a reduction in anaphylaxis frequency [31]. Notably, one study reported that 75% of patients achieved full tolerance during later oral food challenges. Initial oral food challenges at 4 months resulted in dietary liberalization for 66.4% of participants, with this figure rising to 95 new foods introduced across 55 patients by the study’s conclusion [30].
Our real-world experience further validates the significance of this treatment approach. Throughout the course of omalizumab therapy, we have observed in our patient a clinically relevant increase in the threshold dose of cow’s milk. This suggests improved tolerance to even trace amounts of milk proteins, commonly found in various food products, and a significant reduction in the risk of anaphylactic reactions, particularly following transcutaneous and oral exposure to cow’s milk proteins. A notable additional benefit has been the alleviation of the emotional and psychological burden experienced by both the patient and his family, which had previously negatively impacted their quality of life. The patient is continuing with the prescribed omalizumab treatment. We plan to explore the initiation of oral allergen immunotherapy with milk. We also intend to conduct an exposure test with baked milk to evaluate the potential for safely reintroducing this form of milk into the patient’s diet.
Conclusions
While individual responses to omalizumab may vary, the accumulating evidence strongly suggests that prolonged use can maintain elevated tolerance thresholds and facilitate dietary expansion, offering transformative potential in the management of pediatric food allergies. While omalizumab demonstrated marked and rapid improvements in asthma control, evidenced by reduced symptoms, enhanced pulmonary function, and decreased airway inflammation, oral tolerance to cow’s milk was not achieved within the initial five months of therapy. This observation underscores the complex and potentially variable responses to omalizumab in patients with co-existing allergic conditions. Despite the lack of immediate food tolerance induction in this case, the significant asthma benefits and the broader evidence supporting omalizumab’s efficacy in both asthma and food allergy warrant continued investigation and exploration of individualized treatment strategies, particularly in the context of pediatric allergic multimorbidity. Further research is needed to optimize treatment duration, explore combination therapies, and identify predictive factors for successful dual-allergy management with biologic agents such as omalizumab.